- Villela, L. V., Stoerkler, T., Labbé, L, Barriault, L. Bismuth-Catalyzed Photoinduced Ligand-to-Metal Charge Transfer (LMCT) Hydrodecarbonylation of Aldehydes Org. Lett., ASAP.

- Hébert, M., Bellavance, G., Barriault, L. Advanced Synthetic Strategies toward Ginkgolide Diterpenoids: Total Synthesis of (±)-Ginkgolide C and Formal Syntheses of (±)-Ginkgolides A and B J. Org. Chem. 2025, 90, 26, 9147–9159.

- Morris, A., O’Brien, T.E., Barriault, L. Photoredox-Catalyzed Hydroalkylation of C(sp3)-H Acids Chem. Eur. J. 2025, 27, e202501148.

- Montserrat, Z., Villela, L. F., Barriault, L. Photoactivated Formal [3 + 2] / [4+2] Cycloaddition of N-Aryl Cyclopropyl and Cyclobutylamines Eur. J. Org. Chem. 2024, 27, e202400558.

- Morris, A. O., Barriault, L. Redox-Neutral Multicatalytic Cerium Photoredox-Enabled Cleavage of O–H Bearing Substrates Chem. Eur. J. 2024, e202400642

- Tibouti, S., Barriault, L. An Overview of Syntheses of Salvinorin A and its Analogues Chem. Cat. Chem 2024, e202400182

- O’Brien, T.E., Morris, A.O., Villela, L. F., Barriault, L. Synthetic Applications of Photochemically Generated Radicals from Protic C(sp3)–H Bonds Chem. Cat. Chem 2023, e202300989

- Tran, H.; McGee, P., Barriault, L. Formal Synthesis of (±)-Salvinorin A via Gold(I) Catalysis Chem. Eur. J. 2023, e202301640

- Barriault, L.; Hébert, M., Bellavance, G. Total Synthesis of Ginkgolide C and Formal Syntheses of Ginkgolides A and B J. Am. Chem. Soc., 2022, 144, 17702-17796.

- Barriault, L.; Tran, H.; Revol, G.; Poyser, A. Divergent and Modular Synthesis of Terpenoid Scaffolds via a Au(I) Catalyzed One-Pot Cascade Angew. Chem. Int. Ed, 2022, 61, e202110575.

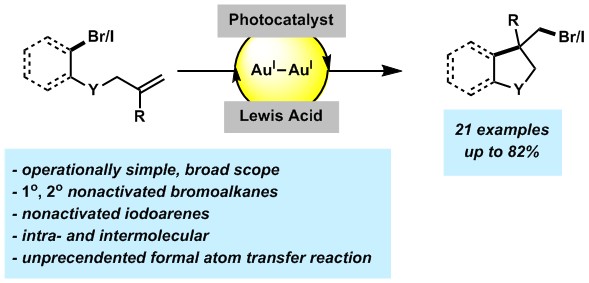
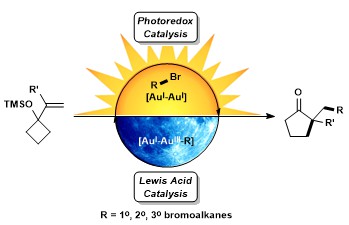
- Barriault, L.; Zidan, M.; McCallum, T.; Swann, R.; Formal Bromine Atom Transfer Radical Addition of Nonactivated Bromoalkanes Using Photoredox Gold Catalysis Org. Lett., 2020, 22, 8401-8406.
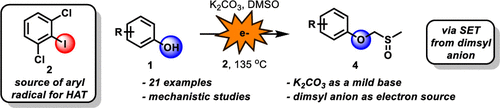
- Barriault, L.; Rohe, S.; Révol, G.; Marmin, T.; Barriault, D.; Barriault, L.; Single-Electron Transfer from Dimsyl Anion in the Alkylation of Phenols J. Org. Chem., 2020, 85, 2806-2813.
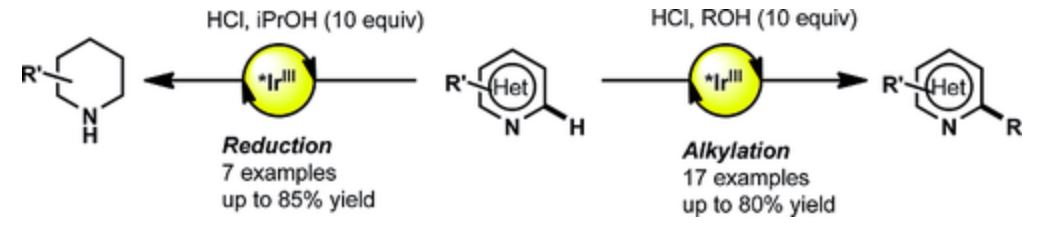
- Barriault, L.; Zidan, M.; Morris, A.O.; McCallum, T.; The Alkylation and Reduction of Heteroarenes with Alcohols Using Photoredox Catalyzed Hydrogen Atom Transfer via Chlorine Atom Generation Eur. J. Org. Chem., 2020, 1453-1458.
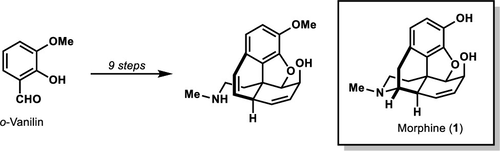
- Barriault, L.; Brousseau, J.; Xiolin A.; A Nine-Step Formal Synthesis of (±)-Morphine Org Lett., 2019, 21, 1347-1349.
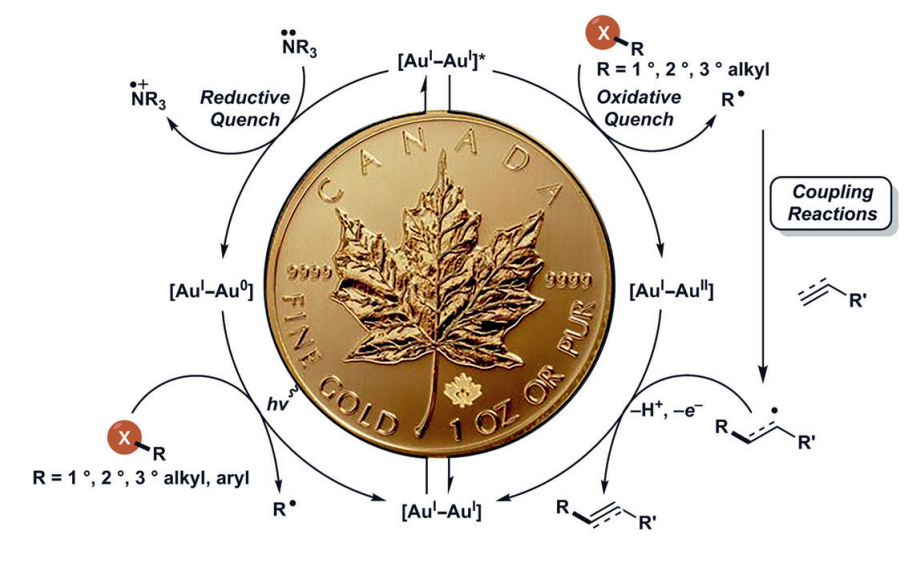
- Barriault, L.; Zidan, M.; Rohe, S.; McCallum, T.; Recent Advances in Mono and Binuclear Gold Photoredox Catalysis Catal. Sci. Technol., 2018, 8, 6019-6028.

- Barriault, L.; Rohe, S.; Morris, A.O.; McCallum, T.; Hydrogen Atom Transfer Reactions via Photoredox Catalyzed Chlorine Atom Generation Angew. Chem, Int. Ed. 2018, 57, 15664-15669.
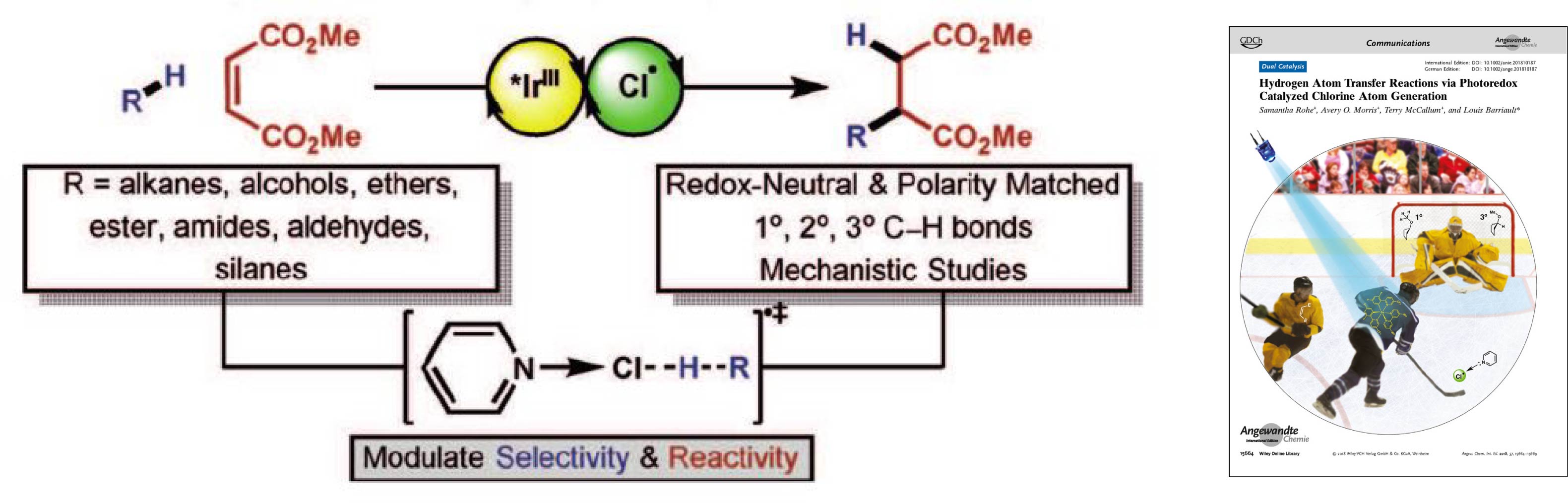
- Barriault, L.; Rohe, S.; McCallum, T.; Morris, A.O., Transformations of Isonitriles with Bromoalkanes using Photoredox Gold Catalysis J. Org. Chem. 2018, 83, 10015-10024
- Barriault, L.; Bellavance, G., Modular Total Syntheses of Hyperforin, Papuaforins A, B and C via Gold(I)-Catalyzed Carbocyclization (Article) J. Org. Chem. 2018, 83, 7215–7230

- Barriault, L.; McGee, P.; Brousseau, J., Development of New Gold (I)-Catalyzed Carbocyclizationsand their Applications in the Synthesis of Natural Products. Isr. J. Chem. 2017, 57, 1-11
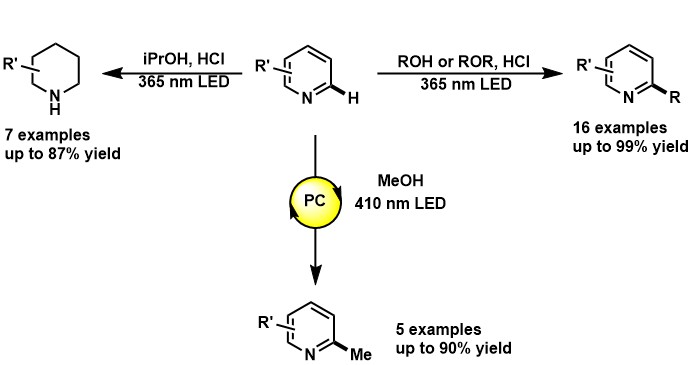
- Barriault, L.; McCallum, T.; Pitre, S. P.; Morin, M.; Scaiano, J. C., The Photochemical Alkylation and Reduction of Heteroarenes. Chemical Science 2017, 8, 7412-7418.
- L. Barriault, M. Zidan, T. McCallum, L. Thai-Savard. Photoredox Meets Gold Lewis Acid Catalysis in the Alkylative Semipinacol Rearrangement: A Photocatalyst with a Dark Side. Org. Chem. Front. 2017, 4, 2092-2096.
- Barriault, L.; McGee, P.; Bétournay, G.; Barabé, F. An 11-Step Total Synthesis of Magellanine through a Gold(I)-Catalyzed Dehydro Diels-Alder Reaction. Angew. Chem. Int. Ed. 2017, 56, 6280–6283.

- McCallum, T.; Rohe, S.; Barriault, L. Thieme Chemistry Journals Awardees – Where Are They Now? What’s Golden: Recent Advances in Organic Transformations Using Photoredox Gold Catalysis. Synlett. 2016, 27, A-Q (account)
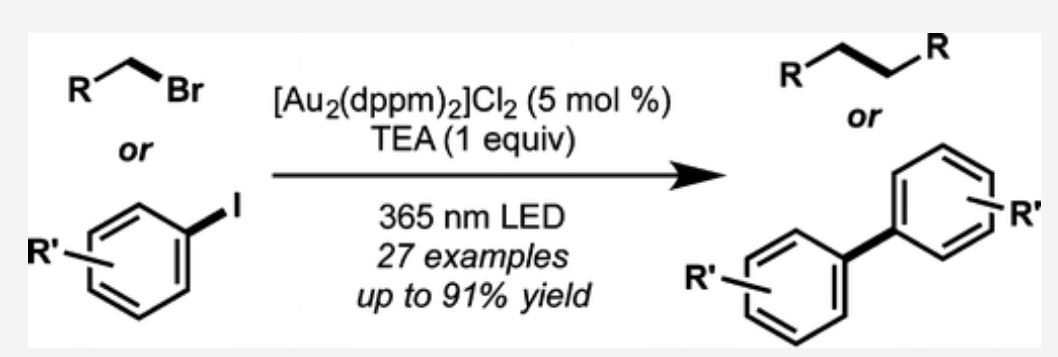
- Barriault, L.; Tran, H.; McCallum, T.; Morin, M. Homocoupling of Iodoarenes and Bromoalkanes Using Photoredox Gold Catalysis: A Light Enabled Au(III) Reductive Elimination. Org Lett. 2016, 18, 4308.
- Barriault, L.; Cannillo, A.; Schwantje, T. R.; Bégin, M., Barabé, F. Gold-Catalyzed Photoredox C(sp2) Cyclization: Formal Synthesis of (±)-Triptolide. Org Lett. 2016, 18, 2592.

- Barriault, L.; McCallum, T. Direct Alkylation of Heteroarenes with Unactivated Bromoalkanes using Photoredox Gold Catalysis. Chem. Sci. 2016, 7, 4754.
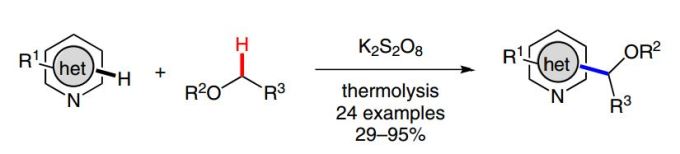
- Barriault, L.; McCallum, T.; Jouanno, L.-A.; Cannillo, A. Persulfate-Enabled Direct C–H Alkylation of Heteroarenes with Unactivated Ethers. Synlett. 2016, 27, 1282.
- Barriault, L.; Scaiano, J. C.; McTiernan, C. D.; Morin, M.; McCallum, T. Polynuclear Gold(I) Complexes in Photoredox Catalysis: Understanding their Reactivity throught Characterization and Kinetic Analysis. Cat. Sci. Technol., 2016, 6, 201.
- Barriault, L.; Kaldas, S. J.; Cannillo, A.; McCallum, T. Indole Functionalization via Photoredox Gold Catalysis. Org Lett. 2015, 17, 2864.

- Barriault, L.; McGee, P.; Bellavance G.; Korobkov, I.; Tarasewicz, A. Synthesis and Isolation of Organogold Complexes via a Controlled 1,2-Silyl Migration. Chem. Eur. J. 2015, 21, 9662.
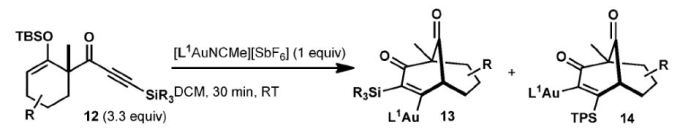
- Barriault, L.; McCallum, T. Light-Enabled Synthesis of Anhydrides and Amides, J. Org. Chem. 2015, 80, 2874.

- Barriault, L.; McCallum, T.; Morin, M.; Slavko, E. Light-Mediated Deoxygenation of Alcohols with a Dimeric Gold Catalyst. Eur. J. Org. Chem. 2015, 81.
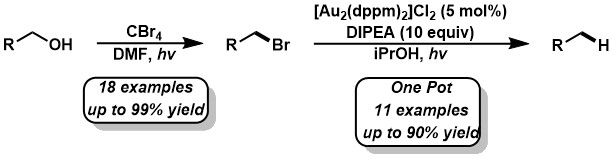
- Barriault, L.; Bellavance, G. Total Syntheses of Hyperforin, Papuaforins A-C and Formal Synthesis of Nemorosone via a Gold(I)-Catalyzed Carbocyclization. Angew. Chem, Int. Ed. 2014, 53, 6701.
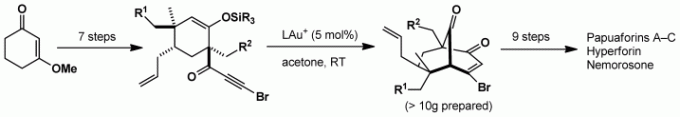
- Barriault, L.; Morin, M.; Levesque, P. Gold(I)-Catalyzed Domino Cyclizations for the Synthesis of Polyaromatic Heterocycles. Beils. J. Org. Chem. 2013, 9, 2625.
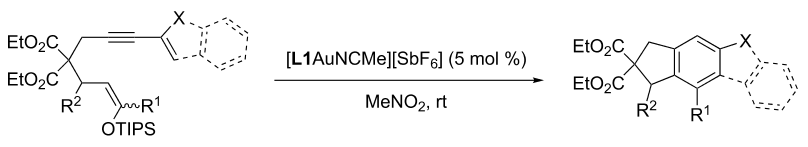
- Barriault, L.; Revol, G.; McCallum, T.; Morin, M.; Gagozs, Photoredox Transformations via Gold Dimeric Complexes. Angew Chem, Int. Ed. 2013, 52, 13342.
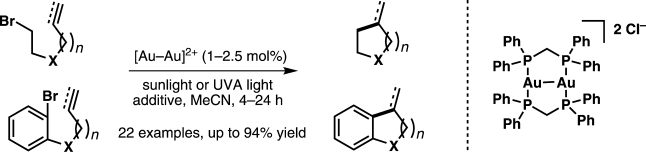
- Barriault, L.; Barabé, F.; Levesque, P.; Bellavance, G.; Sow, B., Gold-Catalyzed Synthesis of Bridged and Fused Carbocyles, Pure and Appl. Chem. 2013, 85, 1161.

- Barriault, L.; Denissova, I,; Goulet, N. Mechanistic Investigation of the Domino Oxy-Cope/Claisen/Ene Reaction and its Application to the Synthesis of Desdimethyl Ambiol B. Synthesis, 2012, 44, 1833. (invited contribution to a special issue on domino reaction, Ed. E. M. Carreira)

- Barriault, L.; Poulin, J, Grisé-Bard, C.M. Formal Synthesis of Vinigrol. Angew. Chem. Int., Ed 2012, 51, 2111. (VIP)

- Barriault, L. ; Barabé, F.; Levesque, P.; Korobkov. I. Synthesis of Fused Carbocycles via a Selective 6-endo dig Gold(I)-Catalyzed Carbocyclization. Org. Let. 2011, 13, 5580.

- Barriault, L. ; Sow, B.; Bellavance, G.; Barabé, F. Concise Synthesis of Bridgehead Ketone via Domino Gold-Catalyzed Cyclizations. Beils. J. Org. Chem. 2011, 7, 1007. (Invited contribution for a special issue in gold-catalyzed process)

- Barriault, L.; Woo. T.; Hooper, J.; Sauer, E. L. O., On the Origin of Altered Diastereomeric Ratios for Anionic versus Neutral Reaction Conditions in the Tandem Oxy-Cope/Claisen/Ene. Eur. Chem. J. 2010, 16, 14124.

- Barriault, L. ; Beingessner, R. ; Farand, J., Toward the Synthesis of Havellocakte. J. Org. Chem. 2010, 75, 6337 (Feature Article)

- Barriault, Barabé, F.; Bétournay, G.; Bellavance G. Gold(I)-Catalyzed Synthesis of Carbon Bridged-Medium Sized Rings. Org. Lett. 2009, 11, 4236. (communication)

- Barriault, Grisé-Bard, C. M.; Poulin J. Pericyclic Domino Reactions : Consise Approaches to Natural Carbocyclic Frameworks. Chem. Rev. Soc. 2009, 38, 3092. (review)
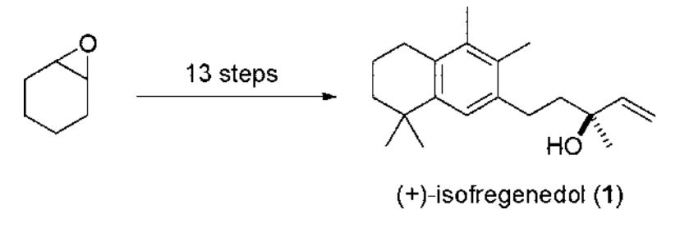
- Barriault, L.; Riou, M. De Novo Synthesis of (+)-Isofregenedol. J. Org. Chem 2008, 73, 7436.
- Barriault, L.; Clément, R.; Grisé, C. M. Stereocontrolled Synthesis of Carbocycles via Four Successive Pericyclic Reactions. Chem. Comm. 2008, 3004. (communication)
- Barriault, L.; Grisé, C. M.; Rodrigue, E. M. Gold(I)-Catalyzed Benzannulation of 3-Hydroxy-1,5-Enynes: An Efficient Synthesis of Substituted Tetrahydronaphthalenes and Related Compounds. Tetrahedron, 2008, 64, 797. (Symposium-in-Print)

- Barriaut, L.; Arns, S.; Lebrun, M. E.; Grisé, C. M.; Denissova, I. Diastereoselective Construction of Quaternary Carbons Via Macrocyclic Conformation. Formal Synthesis of (-)-Mesembrine. J. Org. Chem. 2007, 72, 9314. (article)

- Barriault, L.; Tessier, G. The Conquest of Vinigrol: Creativity, Frustrations and Hope. Org. Prep. Proc. Int. 2007, 39, 311. (review)
- Barriault, L.; Grisé, C. M.; Tessier, G. Synthesis of the Tricyclic Core of Vinigrol via Intramolecular Diels-Alder Reaction. Org. Lett. 2007, 9, 1545. (communication)

- Barriault, L.; Arns, S. Cascading Pericyclic Reactions: Building Complex Carbon Frameworks For Natural Product Synthesis. Chem. Comm. 2007, 2211. (feature article)
- Barriault, L.; Sauer, E. L. O.; Hooper, J.; Woo, T. The Origin of Diastereoselectivity in the Tandem Oxy-Cope/Claisen/Ene Reaction: Experimental and Theoretical Studies of the Ring Inversion Mechanism. J. Am. Chem. Soc. 2007, 129, 2112. (article)

- Barriault, L.; Grisé, C. M. Gold-Catalyzed Synthesis of Substituted Tetrahydronaphthalenes Org. Lett. 2006, 8, 5905. (communication)

- Barriault, L.; Arns, S. A Concise Synthesis of the Neo-Clerodane Skeleton of Teucrolivin A Using An Oxy-Cope/Claisen/Ene Cascade. J. Org. Chem. 2006, 71, 1809. (article)

- Barriault, L.; Lavigne, R. M. A.; Riou, M.; Girardin, M.; Morency, L. Synthesis of Highly Functionalized Bicyclo[m.n.1]alkanones via a Cationic Reaction Cascade. Org. Lett. 2005, 7, 5921. (communication)

- Barriault, L.; Warrington, J. M. Synthesis of the C7-C15 trans Decalin Portion of Natural Antibiotic Tetrodecamycin. Org. Lett. 2005, 7, 4589. (communication)

- Barriault, L.; Morency, L. Studies Toward the Total Synthesis of Vinigrol. Synthesis of the Octalin Ring. J. Org. Chem. 2005, 70, 8841. (article)

- Barriault, L.; Gauvreau, D. Conservation of Planar Chirality in the Tandem Oxy-Cope/Ene Reaction. J. Org. Chem. 2005, 70, 1382. (article)

- Barriault, L.; Sauer, E. L. O. Stereoselective Synthesis of Quaternary Stereocenters: Asymmetric Cross-Coupling and Heck Reactions, 2005, 185-204. Book Chapter (invited contribution).
- Barriault, L.; Sauer, E. L. O. Studies Toward the Synthesis of Wiedemannic acid. Org. Lett. 2004, 6, 3329. (communication)

- Barriault, L.; Morency, L. Stereoselective Synthesis of the cis-Decalin Subunit of Vinigrol via a Three Pericyclic Reactions in Cascade. Tetrahedron Lett. 2004, 45, 6105. (communication)

- Barriault, L; Sauer, E. L. O. Highly Efficient Transfer of Chirality from Macrocyclic Conformation in the Tandem Oxy-Cope/ Claisen/Ene Reaction. J. Am . Soc. Chem. 2004, 126, 8569. (article)

- Barriault, L.; Ang, P. A. J.; Lavigne, R. M. A. Rapid Assembly of the Bicyclo[5.3.1]undecenone Core of Penostatin F: a Successive Diels-Alder/Claisen Reaction Strategy with an Efficient Stereochemical Relay. Org. Lett. 2004, 6, 1317. (communication)

- Barriault, L.; Farand, J. A.; Denissova, I. Microwave Assisted Pericyclic Reaction in Cascade to Construct Decalin Frameworks Possesing Quaternary Centers. Scope and Limitations. Heterocycles 2004, 62, 735. (article) (invited contribution honoring Leo Paquette).
- Barriault, L.; Denissova, I. Stereospecific Synthesis of Quaternary Carbon Centers and Related Functions. Tetrahedron 2003, 59, 10105. (review)

- Barriault, L; Thomas, J.D.; Clément, R. Highly Stereoselctive Hydroxy-Directed Diels-Alder Reaction. J. Org. Chem. 2003, 68, 2317. (article)

- Barriault, L.; Denissova, I. Highly Diastereoselective synthesis of Decalin Skeletons with Quaternary Carbon Centers Using the Tandem Oxy-Cope/Ene/Claisen Reaction. Org.Lett. 2002, 4, 1371. (communication)

- Barriault, L.; Deon, D.H. Total Synthesis of (+)-Arteannuin M Using the Tandem Oxy-Cope/Ene Reaction. Org. Lett. 2001, 3, 1925. (communication)

- Fortin, S.; Barriault, L.; Dory, Y. L.; Deslongchamps,P. Transannular Diels-Alder Reaction of a TTC Macrocycle Having an Activating Dienophile. An Enantioselective Synthesis of ABC [6.6.6] Trans-Syn-Trans and Trans-Anti-Cis Tricycles. J. Am .Chem. Soc. 2001, 123, 8210. (article)
- Barriault, L.; Warrington, J. M. and Yap, G. P. A. Tandem oxy-Cope/Ene Reaction of 1,2-Divinylcyclohexanols. Org. Lett. 2000, 2, 663. (communication)

- Paquette, L. A.; Barriault, L.; Pissarnitski, D.; Johnston, J. N. Stereocontrolled Elaboration of Natural (-)-Polycavernoside A, a Powerfully Toxic Metabolite of the Red Alga Polycavernosa Tsudai. J. Am. Chem. Soc. 2000, 122, 619. (article)

- Barriault, L.; Boulet, S. L.; Fujiwara, K.; Murai, A.; Paquette, L. A.; Yotsu-Yamashita M. Synthesis and Biological Evaluation of Analogues of the Marine Toxin Polycavernoside A. Bioorg. Medicin. Chem. Lett. 1999, 9, 2069. (communication)
- Barriault, L.; Paquette, L. A. The first Total Syntheses of Ircinol A, Ircinal A and Manzamines A and D. Chemtracts 1999, 12, 276. (review)
- Paquette, L. A.; Barriault, L.; Pissarnitski, D. A Convergent Total Synthesis of the Macrolactone Disacharide Toxin (-)- Polycavernoside A. J. Am. Chem. Soc. 1999, 121, 4542. (communication)
- Paquette, L. A., Pissarnitski, D.; Barriault, L.A Modular Enantioselective Approach to Construction of the Macrolactone Core of Polycavernoside A. J. Org. Chem. 1998, 63, 7389. (article)

- Barriault, L.; Deslongchamps, P. Synthesis of 17-Membered Ring Macrocyle: Studies Toward the Enantioselective Synthesis of Fusidic Acid. Bull. Soc. Chim. Fr. 1997, 134, 969. (article)
- Barriault, L.; Ouellet, S. G.; Deslongchamps, P. Transannular Diels-Alder reaction Studies with an Activated Dienophile. An Enantioselective Synthesis of an ABC [6.6.6] Trans-Syn-Cis Tricycle. Tetrahedron 1997, 53, 14937. (article)